Product Description
Perindopril is used alone or in combination with other medications to treat high blood pressure. Perindopril is in a class of medications called angiotensin-converting enzyme (ACE) inhibitors. It makes blood flow more smoothly by preventing the production of certain natural chemicals that tighten the blood vessels. (Sourced from: https://medlineplus.gov/druginfo/meds/a602017.html)
Mechanisms of Action: ACE Inhibitor
Novel Mechanism: No
Modality: Small Molecule
Route of Administration: Oral
FDA Designation: *
Approval Status: Approved
Approved Countries: Algeria | Argentina | Australia | Austria | Bangladesh | Belgium | Bosnia | Brazil | Bulgaria | Canada | Chile | Colombia | Croatia | Cyprus | Czech | Denmark | Dominican Republic | Ecuador | Egypt | Estonia | Finland | France | Germany | Greece | Hong Kong | Hungary | Iceland | India | Indonesia | Ireland | Italy | Jordan | Korea | Latvia | Lebanon | Lithuania | Luxembourg | Malaysia | Malta | Mexico | Morocco | Netherlands | New Zealand | Norway | Pakistan | Peru | Philippines | Poland | Portugal | Romania | Russia | Saudi Arabia | Serbia | Singapore | Slovakia | Slovenia | South Africa | Spain | Sri Lanka | Sweden | Switzerland | Taiwan | Thailand | Tunisia | Turkey | Ukraine | United Arab Emirates | United Kingdom | United States | Uruguay | Venezuela | Vietnam
Approved Indications: None
Known Adverse Events: None
Company: Servier Russia
Company Location:
Company Founding Year: None
Additional Commercial Interests: None
Clinical Description
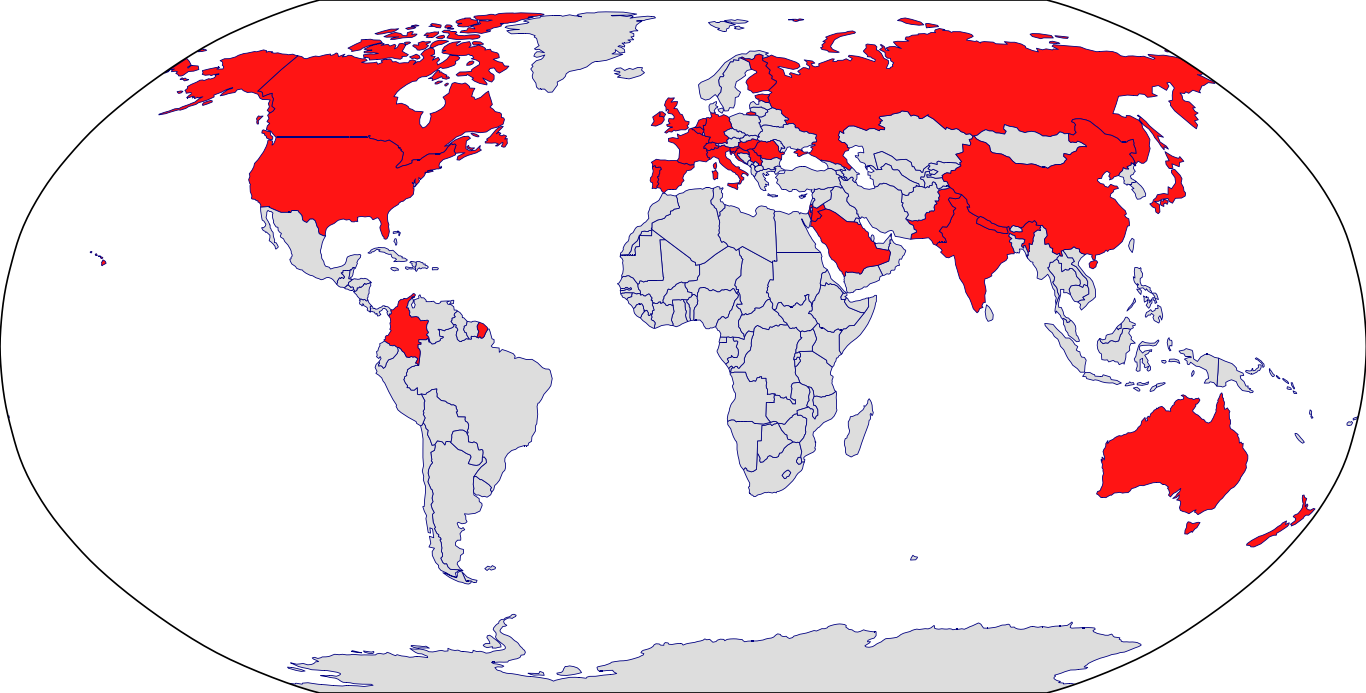
Countries in Clinic: Australia, Belgium, Canada, Colombia, Croatia, Czech Republic, Estonia, Finland, France, Germany, Hungary, India, Ireland, Israel, Italy, Japan, Nepal, Netherlands, New Zealand, Pakistan, Portugal, Romania, Saudi Arabia, Serbia, Slovenia, Spain, Switzerland, United Kingdom, United States
Active Clinical Trial Count: 3
Recent & Upcoming Milestones
Highest Development Phases
Phase 3: COVID-19|Hypertension|Influenza, Human|Pneumonia
Phase 1: Osteoporosis, Postmenopausal
Trial ID |
Trial |
Phase |
Trial Status |
Disease |
Primary Completion Date |
Probability of Success |
Latest Trial Update Date |
Data Updated |
|---|---|---|---|---|---|---|---|---|
ACTRN12621001077853p |
2006-7041-83/hah | P1 |
Not yet recruiting |
Hypertension|Osteoporosis, Postmenopausal |
2022-02-13 |
|||
NCT02735707 |
REMAP-CAP | P3 |
Recruiting |
COVID-19|Pneumonia|Influenza, Human |
2026-02-01 |
2024-11-27 |
||
2020-004891-16 |
2020-004891-16 | P3 |
Completed |
Hypertension |
2023-12-14 |
2025-07-09 |
Treatments |
Recent News Events
Date |
Type |
Title |
|---|---|---|
|
02/21/2024 |
News Article |
Discover this new generic drug in the PERINDOPRIL range |
|
09/09/2023 |
News Article |
The Cumulative Impact of COVID-19 on Military Battery Packs Market disrupted market Size, market Share along with market Growth Rate of 5.8% CAGR from 2023 to 2030 |
|
09/05/2023 |
News Article |
Pharmascience Canada launches a new generic drug to treat hypertension |
|
07/31/2023 |
News Article |
Bioxytran Names Leslie Ajayi as Interim CMO and Head of the Medical Advisory Board |


