Product Description
Mercaptopurine is a medication used in the management and treatment of acute lymphoblastic leukemia. It is in the class of medications known as purine antagonists. (Sourced from: https://www.ncbi.nlm.nih.gov/books/NBK557620/)
Mechanisms of Action: Immunosuppressive
Novel Mechanism: No
Modality: Small Molecule
Route of Administration: Oral
FDA Designation: *
Approval Status: Approved
Approved Countries: Algeria | Argentina | Australia | Austria | Bangladesh | Belgium | Brazil | Bulgaria | Canada | Chile | Colombia | Croatia | Czech | Denmark | Dominican Republic | Ecuador | Egypt | Estonia | European Medicines Agency | Finland | France | Germany | Greece | Hong Kong | Hungary | Iceland | India | Ireland | Israel | Italy | Jordan | Korea | Latvia | Lebanon | Lithuania | Luxembourg | Malta | Mexico | Morocco | Netherlands | New Zealand | Norway | Pakistan | Peru | Philippines | Poland | Portugal | Romania | Russia | Saudi Arabia | Singapore | Slovakia | Slovenia | Spain | Sri Lanka | Sweden | Switzerland | Taiwan | Thailand | Tunisia | Turkey | Ukraine | United Arab Emirates | United Kingdom | United States | Uruguay | Vietnam
Approved Indications: None
Known Adverse Events: None
Company: Teva
Company Location: Europe
Company Founding Year: 1901
Additional Commercial Interests: None
Clinical Description
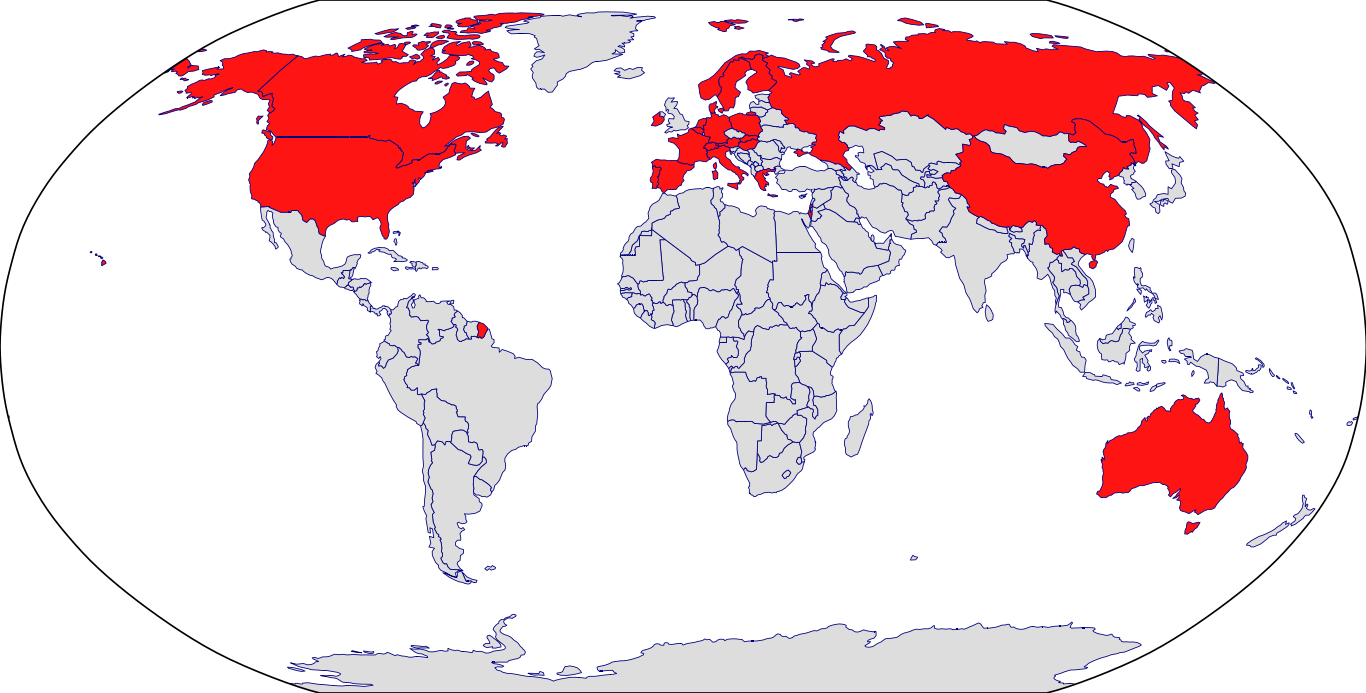
Countries in Clinic: Australia, Austria, Belgium, Canada, Czech Republic, Denmark, Greece, Ireland, Italy, Netherlands, Norway, Poland, Spain, Sweden, United States
Active Clinical Trial Count: 8
Recent & Upcoming Milestones
Highest Development Phases
Phase 3: Acute Lymphoid Leukemia|Colitis, Ulcerative|Histiocytosis|Precursor Cell Lymphoblastic Leukemia-Lymphoma
Phase 2: Lymphoid Leukemia
Trial ID |
Trial |
Phase |
Trial Status |
Disease |
Primary Completion Date |
Probability of Success |
Latest Trial Update Date |
Data Updated |
|---|---|---|---|---|---|---|---|---|
NCT05848687 |
TINI 2 | P2 |
Recruiting |
Precursor Cell Lymphoblastic Leukemia-Lymphoma|Lymphoid Leukemia |
2028-12-01 |
12% |
2024-11-27 |
Primary Endpoints|Treatments |
NCT03117751 |
TOT17 | P3 |
Active, not recruiting |
Acute Lymphoid Leukemia|Precursor Cell Lymphoblastic Leukemia-Lymphoma |
2026-09-30 |
10% |
2024-11-27 |
Primary Endpoints|Treatments |
2024-512676-36-00 |
042011 | P3 |
Not yet recruiting |
Histiocytosis |
2025-12-30 |
2025-05-02 |
Treatments |
|
2015-005260-41 |
OPTIC | P3 |
Active, not recruiting |
Colitis, Ulcerative |
2019-10-13 |
2025-05-17 |
Treatments |
|
2016-003568-38 |
2016-003568-38 | P3 |
Active, not recruiting |
Histiocytosis |
None |
2025-05-11 |
Treatments |
|
NCT06533748 |
SJALL23H | P2 |
Recruiting |
Acute Lymphoid Leukemia|Precursor Cell Lymphoblastic Leukemia-Lymphoma |
2028-05-01 |
12% |
2025-01-11 |
|
NCT03020030 |
NCT03020030 | P3 |
Active, not recruiting |
Precursor Cell Lymphoblastic Leukemia-Lymphoma|Acute Lymphoid Leukemia |
2026-11-30 |
40% |
2025-01-04 |
|
2012-000793-30 |
IntReALL SR 2010 | P3 |
Completed |
Acute Lymphoid Leukemia |
2024-04-25 |
2025-05-06 |
Treatments |


